Sanitizing Food Equipment and Manufacturing Areas with Alcohol and Chlorine
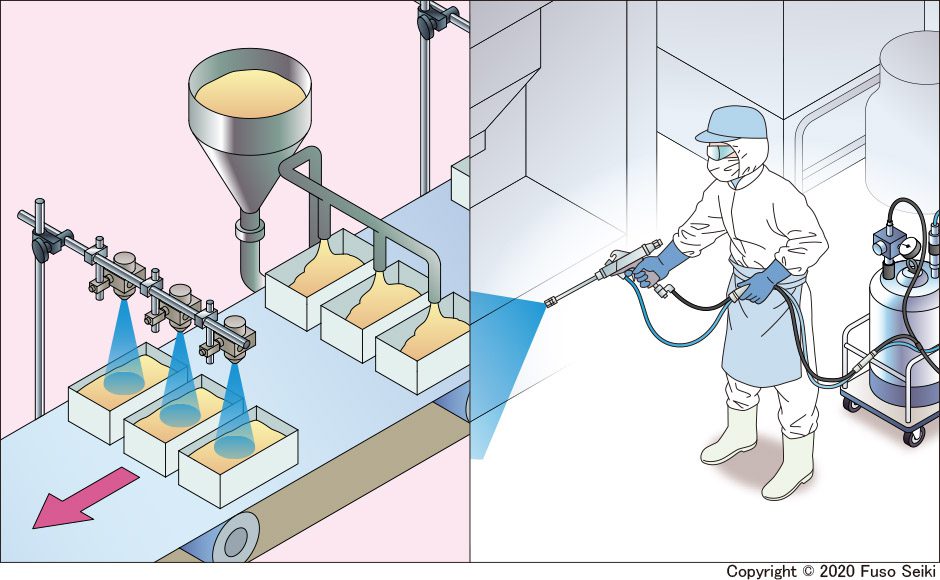
COVID-19 is rapidly spreading around the world. Two preventive measures recommended by the World Health Organization (WHO) include sanitization with alcohol or sodium hypochlorite. This column introduces methods of spray-based disinfection used in a variety of areas, including food manufacturing facilities.
Ethanol and Chlorine Kill Viruses and Microbes
As of March 2020, COVID-19 is continuing to spread from China to the rest of the world. As with every year, this winter has seen large numbers of people infected with other potentially fatal diseases such as influenza and norovirus. Alongside measures such as handwashing, sanitization using alcohol and chlorine can also help prevent the spread of infectious diseases.
The WHO’s interim guidance for COVID-19 was announced in February 2020. The guidance mentions that a 0.1% solution of sodium hypochlorite, or a solution of 62–71% ethanol, may be highly effective at deactivating novel coronavirus in the same way as other enveloped viruses.
Alcohols such as ethanol destroy the lipid-based cell membranes of bacteria and envelopes (outer coatings) of viruses, killing (deactivating) them in the process. Their minimal effects on the human body have led to them commonly being used as hand sanitizers.
Chlorine-based disinfectants such as sodium hypochlorite and hypochlorous acid water have strong oxidizing effects. This allows them to destroy the proteins that bacteria and viruses consist of. These substances are different from ethanol, in that they are effective against viruses without envelopes such as norovirus.
However, at high concentrations, sodium hypochlorite is used as bleach. Such a powerful chemical cannot be used to disinfect parts of the body like the hands. Chemicals such as these are primarily used for sanitizing water and sewerage, as well as utensils at food manufacturing facilities.
Using Spray Guns to Sanitize Food Before Wrapping
Let’s now examine the use of spray guns to distribute disinfectants.
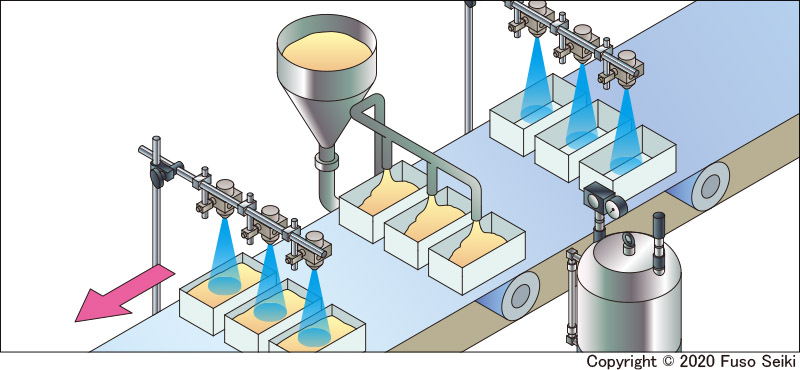
One popular usage method is alcohol-based sterilization at food manufacturing facilities. Short sprays of alcohol are used to disinfect items such as margarine, eclairs, and sausages directly before they are packed. As well as its antimicrobial properties, alcohol can also be used to help prevent food from changing color, or sprayed on equipment such as conveyor belts.
Some odor-producing facilities, such as aquarium pools and pig farms, have installed automatic spray guns at high locations. These guns are used to spray slightly acidic hypochlorous acid water (an aqueous solution produced by electrolyzing hydrochloric acid or a solution of sodium chloride) for deodorizing purposes.
Slightly acidic hypochlorous acid water is a different solution from sodium hypochlorite. Japan’s Ministry of Health, Labour and Welfare has certified hypochlorous acid water as having the same sanitizing effects as sodium hypochlorite. A risk assessment report of additives published by the Food Safety Commission of Japan in 2012 stated that, when orally administered to mice in experiments, slightly acidic hypochlorous acid water did not display any problematic toxic or carcinogenic properties.
Two points of difficulty include the relatively high cost of production equipment and the substance’s chemical instability, which leads to spontaneous decomposition during storage. However, its strong sanitizing effects and safety mean that slightly acidic hypochlorous acid water is highly valued as a disinfectant.
Mobile Cleaning of Facilities Using Handheld Spray Guns
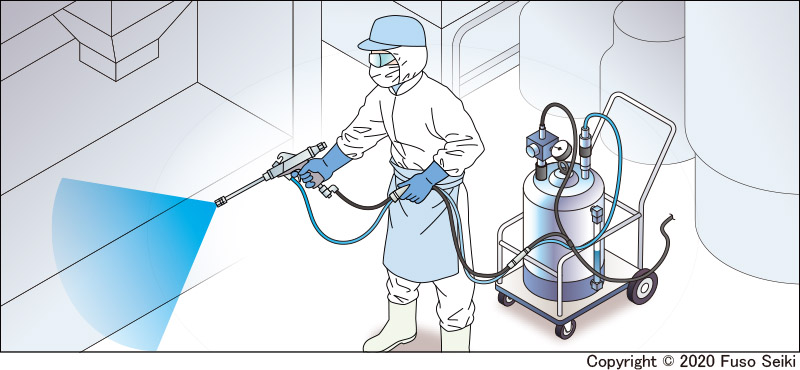
Facilities can also be cleaned using handheld spray guns. The pressure feed tanks attached to the guns can be filled with a solution of sodium hypochlorite or slightly acidic hypochlorous acid water, which can then be used by workers to clean floors and equipment.
Caution: Sodium Hypochlorite May Cause Metal to Corrode
When using sodium hypochlorite, caution is needed as it may cause metals to corrode. Please be sure to confirm the material properties of the object being sanitized, as well as that of the spray gun and the pressure feed tank.
Standard Lumina automatic spray guns feature chrome-plated brass and may therefore be corroded. Please be sure to use an all stainless steel model (a 303 stainless steel model, e.g. an STS model) or a polyphenylene sulfide (PPS) resin model (HMP series). However, when used for long periods of time, even a 303 stainless steel model may be corroded. A PPS resin model will not.
To prevent corrosion, immediately after use, please remove any solution from the spray gun and from the stainless steel tank, and allow them to dry. Once it dries, sodium hypochlorite evaporates as gas, leaving no residue behind. However, if it is not allowed to dry completely and some of the solution remains, it may cause corrosion.
Therefore, after use, it would be best to wipe down the spray gun and tank with water and to run water through the inlets. It is also standard practice to wipe down with water or rinse the object that has been sanitized, ten minutes after sanitization is complete.
Alcohol and slightly acidic hypochlorous acid water are only slightly corrosive. However, ethanol and other alcohols may cause aluminum to corrode, so caution is necessary.
This article has introduced several examples of spray-based sanitization. However, Lumina (Fuso Seiki Co., Ltd.) cannot guarantee the scientific efficacy of their sanitizing effects. Lumina cannot take any responsibility for any health hazards or damage to property arising from the spraying of disinfectants. When introducing these methods, ensure that you accurately confirm any conditions related to their use, then operate them with due care.

